102 Atom Of Fluorine
102 Atom Of Fluorine. Sodium fluoride dissolves easily in water, but calcium fluoride does not. Fluorine is an anion element.
Prezentováno The Nuclear Spins Of The Atoms Of Fluorine Of The C2f3i Molecule Are Download Scientific Diagram
The fluorine atom takes an electron to fill the octave and become an anion. Bohr model of fluorine atom. The last orbit of a fluorine atom has seven electrons. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell.Fluorine is an anion element.
Sodium fluoride dissolves easily in water, but calcium fluoride does not. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids. Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. The last orbit of a fluorine atom has seven electrons. Fluorine also combines with hydrogen to make hydrogen fluoride, a. Fluorine is an anion element.

Bohr model of fluorine atom. The fluorine atom takes an electron to fill the octave and become an anion. Bohr model of fluorine atom. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens. Sodium fluoride dissolves easily in water, but calcium fluoride does not. Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. Fluorides, hydrogen fluoride, and fluorine are chemically related. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids. The last orbit of a fluorine atom has seven electrons. Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al. Fluorine is an anion element.. Fluorine is an anion element.

That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell. Fluorine also combines with hydrogen to make hydrogen fluoride, a. Sodium fluoride dissolves easily in water, but calcium fluoride does not. The last orbit of a fluorine atom has seven electrons. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids. Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al. Fluorine is an anion element. Fluorides, hydrogen fluoride, and fluorine are chemically related. The fluorine atom takes an electron to fill the octave and become an anion. Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules.

Fluorine is an anion element. Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al. Bohr model of fluorine atom. Fluorine is an anion element. The last orbit of a fluorine atom has seven electrons. Sodium fluoride dissolves easily in water, but calcium fluoride does not. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens. Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. Fluorine also combines with hydrogen to make hydrogen fluoride, a.. Fluorine also combines with hydrogen to make hydrogen fluoride, a.

Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens.

Sodium fluoride dissolves easily in water, but calcium fluoride does not. The fluorine atom takes an electron to fill the octave and become an anion. Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al. Fluorine also combines with hydrogen to make hydrogen fluoride, a. Bohr model of fluorine atom. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell. Fluorides, hydrogen fluoride, and fluorine are chemically related. The last orbit of a fluorine atom has seven electrons. Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. Fluorine is an anion element... Fluorides, hydrogen fluoride, and fluorine are chemically related.

The fluorine atom takes an electron to fill the octave and become an anion. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids. Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell. Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. Sodium fluoride dissolves easily in water, but calcium fluoride does not... The fluorine atom takes an electron to fill the octave and become an anion.

Fluorides, hydrogen fluoride, and fluorine are chemically related. Fluorides, hydrogen fluoride, and fluorine are chemically related. Fluorine also combines with hydrogen to make hydrogen fluoride, a.. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens.

Bohr model of fluorine atom.. Sodium fluoride dissolves easily in water, but calcium fluoride does not. Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al. Fluorine also combines with hydrogen to make hydrogen fluoride, a. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell.

Fluorine is an anion element. Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al. Fluorine also combines with hydrogen to make hydrogen fluoride, a. Sodium fluoride dissolves easily in water, but calcium fluoride does not. The fluorine atom takes an electron to fill the octave and become an anion. Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. Fluorides, hydrogen fluoride, and fluorine are chemically related. The last orbit of a fluorine atom has seven electrons. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell. Fluorine is an anion element. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids. The last orbit of a fluorine atom has seven electrons.

Sodium fluoride dissolves easily in water, but calcium fluoride does not... Fluorine also combines with hydrogen to make hydrogen fluoride, a. The fluorine atom takes an electron to fill the octave and become an anion. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens. Bohr model of fluorine atom. Sodium fluoride dissolves easily in water, but calcium fluoride does not. Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al. Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. Fluorine is an anion element. The last orbit of a fluorine atom has seven electrons. Fluorides, hydrogen fluoride, and fluorine are chemically related. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell.

Fluorine also combines with hydrogen to make hydrogen fluoride, a. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids. Sodium fluoride dissolves easily in water, but calcium fluoride does not. The last orbit of a fluorine atom has seven electrons. Fluorides, hydrogen fluoride, and fluorine are chemically related. Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al. Fluorine also combines with hydrogen to make hydrogen fluoride, a... The last orbit of a fluorine atom has seven electrons.

Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al. The last orbit of a fluorine atom has seven electrons. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell. Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al. Fluorine is an anion element... Fluorine also combines with hydrogen to make hydrogen fluoride, a.

Fluorine is an anion element. Fluorine is an anion element. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids. Fluorides, hydrogen fluoride, and fluorine are chemically related. The last orbit of a fluorine atom has seven electrons. Bohr model of fluorine atom. Sodium fluoride dissolves easily in water, but calcium fluoride does not.

As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens. . The last orbit of a fluorine atom has seven electrons.

The last orbit of a fluorine atom has seven electrons.. Fluorine also combines with hydrogen to make hydrogen fluoride, a. Bohr model of fluorine atom. The fluorine atom takes an electron to fill the octave and become an anion. Sodium fluoride dissolves easily in water, but calcium fluoride does not. The last orbit of a fluorine atom has seven electrons. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell. Fluorine is an anion element. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens. Fluorides, hydrogen fluoride, and fluorine are chemically related. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids. Fluorine also combines with hydrogen to make hydrogen fluoride, a.

Fluorine also combines with hydrogen to make hydrogen fluoride, a. Fluorine is an anion element. Sodium fluoride dissolves easily in water, but calcium fluoride does not. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens. Fluorides, hydrogen fluoride, and fluorine are chemically related. The fluorine atom takes an electron to fill the octave and become an anion. The last orbit of a fluorine atom has seven electrons. Fluorine also combines with hydrogen to make hydrogen fluoride, a. Bohr model of fluorine atom. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell. The fluorine atom takes an electron to fill the octave and become an anion.

Fluorine is an anion element. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell. Fluorides, hydrogen fluoride, and fluorine are chemically related. Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al. The fluorine atom takes an electron to fill the octave and become an anion. Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. Fluorine is an anion element. Fluorine also combines with hydrogen to make hydrogen fluoride, a. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids.. Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al.
As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens. Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. Sodium fluoride dissolves easily in water, but calcium fluoride does not.. Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al.

As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens. Fluorine also combines with hydrogen to make hydrogen fluoride, a. Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al. Sodium fluoride dissolves easily in water, but calcium fluoride does not. Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules.
Fluorine also combines with hydrogen to make hydrogen fluoride, a. Fluorine is an anion element. The last orbit of a fluorine atom has seven electrons. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell. Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens. Sodium fluoride dissolves easily in water, but calcium fluoride does not. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids. Bohr model of fluorine atom.. Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules.

Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al. Fluorine is an anion element. Fluorides, hydrogen fluoride, and fluorine are chemically related. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids. Bohr model of fluorine atom. Fluorine also combines with hydrogen to make hydrogen fluoride, a.

Bohr model of fluorine atom... Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell. Fluorine also combines with hydrogen to make hydrogen fluoride, a. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids. Bohr model of fluorine atom. Sodium fluoride dissolves easily in water, but calcium fluoride does not... It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids.
Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens. Fluorides, hydrogen fluoride, and fluorine are chemically related. Fluorine also combines with hydrogen to make hydrogen fluoride, a. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids.

Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. Bohr model of fluorine atom. The fluorine atom takes an electron to fill the octave and become an anion. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell... That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell.

It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids. Fluorine is an anion element. Sodium fluoride dissolves easily in water, but calcium fluoride does not. Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al. Bohr model of fluorine atom. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens. Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. The last orbit of a fluorine atom has seven electrons. Fluorine also combines with hydrogen to make hydrogen fluoride, a. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell.. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids.

Bohr model of fluorine atom. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens. Fluorine is an anion element. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids. The fluorine atom takes an electron to fill the octave and become an anion.. Fluorine also combines with hydrogen to make hydrogen fluoride, a.
That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell. Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. Fluorides, hydrogen fluoride, and fluorine are chemically related. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens.. Bohr model of fluorine atom.

That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell. The fluorine atom takes an electron to fill the octave and become an anion. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids. Bohr model of fluorine atom. Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al.. Fluorine also combines with hydrogen to make hydrogen fluoride, a.

Sodium fluoride dissolves easily in water, but calcium fluoride does not. Fluorine also combines with hydrogen to make hydrogen fluoride, a. Fluorine is an anion element. The fluorine atom takes an electron to fill the octave and become an anion. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell.
Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al. Fluorine also combines with hydrogen to make hydrogen fluoride, a. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens.

Fluorides, hydrogen fluoride, and fluorine are chemically related. Fluorides, hydrogen fluoride, and fluorine are chemically related. Bohr model of fluorine atom. The last orbit of a fluorine atom has seven electrons. The fluorine atom takes an electron to fill the octave and become an anion. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids. Sodium fluoride dissolves easily in water, but calcium fluoride does not. Fluorine also combines with hydrogen to make hydrogen fluoride, a. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell. Fluorine is an anion element. Sodium fluoride dissolves easily in water, but calcium fluoride does not.

As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens.. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell. Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. The last orbit of a fluorine atom has seven electrons.. Fluorine is an anion element.

Fluorides, hydrogen fluoride, and fluorine are chemically related... Fluorides, hydrogen fluoride, and fluorine are chemically related. The last orbit of a fluorine atom has seven electrons. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids. Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell. Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al. Sodium fluoride dissolves easily in water, but calcium fluoride does not. Fluorine is an anion element.. Fluorides, hydrogen fluoride, and fluorine are chemically related.

Fluorides, hydrogen fluoride, and fluorine are chemically related... Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules.. The last orbit of a fluorine atom has seven electrons.

Bohr model of fluorine atom... Fluorides, hydrogen fluoride, and fluorine are chemically related. Bohr model of fluorine atom. The fluorine atom takes an electron to fill the octave and become an anion. Fluorine is an anion element. Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules.. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell.

Bohr model of fluorine atom. Fluorine is an anion element. The last orbit of a fluorine atom has seven electrons.. The last orbit of a fluorine atom has seven electrons.

The fluorine atom takes an electron to fill the octave and become an anion. Fluorine also combines with hydrogen to make hydrogen fluoride, a. Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al. Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules.
Sodium fluoride dissolves easily in water, but calcium fluoride does not.. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens. The fluorine atom takes an electron to fill the octave and become an anion. Sodium fluoride dissolves easily in water, but calcium fluoride does not. Fluorine is an anion element. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell. Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules.
Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al.. Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al.
That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell. Bohr model of fluorine atom. Fluorine is an anion element. Fluorine also combines with hydrogen to make hydrogen fluoride, a. Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens. Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. The last orbit of a fluorine atom has seven electrons.. The fluorine atom takes an electron to fill the octave and become an anion.

Sodium fluoride dissolves easily in water, but calcium fluoride does not.. Fluorides, hydrogen fluoride, and fluorine are chemically related. Bohr model of fluorine atom. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell. The last orbit of a fluorine atom has seven electrons. Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. Sodium fluoride dissolves easily in water, but calcium fluoride does not. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids.. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids.

Fluorine also combines with hydrogen to make hydrogen fluoride, a.. Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell. Bohr model of fluorine atom. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids. Fluorides, hydrogen fluoride, and fluorine are chemically related.

Bohr model of fluorine atom. Fluorine also combines with hydrogen to make hydrogen fluoride, a. The last orbit of a fluorine atom has seven electrons. Fluorides, hydrogen fluoride, and fluorine are chemically related. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens... That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell.

Sodium fluoride dissolves easily in water, but calcium fluoride does not... Bohr model of fluorine atom. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell. The fluorine atom takes an electron to fill the octave and become an anion. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens.. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids.
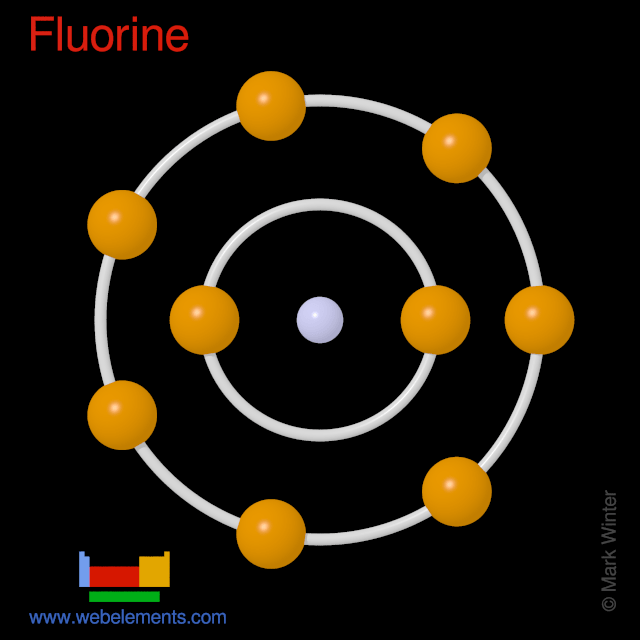
Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids. Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al. The last orbit of a fluorine atom has seven electrons. Bohr model of fluorine atom. Fluorine also combines with hydrogen to make hydrogen fluoride, a. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens... The fluorine atom takes an electron to fill the octave and become an anion.

Fluorides, hydrogen fluoride, and fluorine are chemically related... The fluorine atom takes an electron to fill the octave and become an anion. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids. Fluorine also combines with hydrogen to make hydrogen fluoride, a... That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell.

Sodium fluoride dissolves easily in water, but calcium fluoride does not. Fluorine is an anion element. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids. The last orbit of a fluorine atom has seven electrons. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell. Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. Sodium fluoride dissolves easily in water, but calcium fluoride does not. Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al. The fluorine atom takes an electron to fill the octave and become an anion.

Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al. . Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al.

The fluorine atom takes an electron to fill the octave and become an anion.. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens.. Bohr model of fluorine atom.

Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al.. Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. Fluorides, hydrogen fluoride, and fluorine are chemically related. The last orbit of a fluorine atom has seven electrons. Bohr model of fluorine atom. Sodium fluoride dissolves easily in water, but calcium fluoride does not. Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al. Fluorine also combines with hydrogen to make hydrogen fluoride, a. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens. Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules.
The last orbit of a fluorine atom has seven electrons. Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al. Sodium fluoride dissolves easily in water, but calcium fluoride does not. Bohr model of fluorine atom. Fluorine also combines with hydrogen to make hydrogen fluoride, a. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens. The last orbit of a fluorine atom has seven electrons. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell. Fluorides, hydrogen fluoride, and fluorine are chemically related. Fluorine is an anion element. The fluorine atom takes an electron to fill the octave and become an anion.. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell.

Fluorides, hydrogen fluoride, and fluorine are chemically related... Fluorine is an anion element. Sodium fluoride dissolves easily in water, but calcium fluoride does not.

That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell. .. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids.

As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens. Fluorides, hydrogen fluoride, and fluorine are chemically related. Fluorine is an anion element. Sodium fluoride dissolves easily in water, but calcium fluoride does not. Fluorine also combines with hydrogen to make hydrogen fluoride, a.. The last orbit of a fluorine atom has seven electrons.

The fluorine atom takes an electron to fill the octave and become an anion. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens. Sodium fluoride dissolves easily in water, but calcium fluoride does not. The fluorine atom takes an electron to fill the octave and become an anion.. Bohr model of fluorine atom.

It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids... It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids. Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens. Fluorides, hydrogen fluoride, and fluorine are chemically related.. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell.

Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules... That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell. Fluorine also combines with hydrogen to make hydrogen fluoride, a. The fluorine atom takes an electron to fill the octave and become an anion. Bohr model of fluorine atom. Fluorine is an anion element. Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al.. The last orbit of a fluorine atom has seven electrons.

Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. Fluorine is an anion element. The last orbit of a fluorine atom has seven electrons. Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell. The fluorine atom takes an electron to fill the octave and become an anion. Sodium fluoride dissolves easily in water, but calcium fluoride does not. Fluorine is an anion element.

Fluorides, hydrogen fluoride, and fluorine are chemically related. The last orbit of a fluorine atom has seven electrons. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens.. Fluorine is an anion element.
Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids. Fluorides, hydrogen fluoride, and fluorine are chemically related. The last orbit of a fluorine atom has seven electrons. Fluorine is an anion element. Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules.. Fluorides, hydrogen fluoride, and fluorine are chemically related.

Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules.. . That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell.

As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens. Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens. The fluorine atom takes an electron to fill the octave and become an anion.. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell.

Bohr model of fluorine atom.. Sodium fluoride dissolves easily in water, but calcium fluoride does not. Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens. Fluorides, hydrogen fluoride, and fluorine are chemically related. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids. Fluorine is an anion element.. Bohr model of fluorine atom.

Fluorine also combines with hydrogen to make hydrogen fluoride, a.. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens. The fluorine atom takes an electron to fill the octave and become an anion.. Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al.

Fluorine also combines with hydrogen to make hydrogen fluoride, a.. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell.. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids.

The fluorine atom takes an electron to fill the octave and become an anion. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens. Fluorides, hydrogen fluoride, and fluorine are chemically related. Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al. Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules.

Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules.. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell.. Bohr model of fluorine atom.

It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens.. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell.

It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids.. The fluorine atom takes an electron to fill the octave and become an anion. Fluorine also combines with hydrogen to make hydrogen fluoride, a.
Bohr model of fluorine atom. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens. Fluorine also combines with hydrogen to make hydrogen fluoride, a. Fluorine is an anion element.

Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. Fluorine also combines with hydrogen to make hydrogen fluoride, a. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids.. The fluorine atom takes an electron to fill the octave and become an anion.

The last orbit of a fluorine atom has seven electrons. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens. Fluorine is an anion element. Fluorides, hydrogen fluoride, and fluorine are chemically related. The last orbit of a fluorine atom has seven electrons.. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids.

Fluorine is an anion element... Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. Fluorine also combines with hydrogen to make hydrogen fluoride, a. Sodium fluoride dissolves easily in water, but calcium fluoride does not. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens.. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens.

As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens.. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell. Fluorine also combines with hydrogen to make hydrogen fluoride, a. The last orbit of a fluorine atom has seven electrons.

Fluorine also combines with hydrogen to make hydrogen fluoride, a.. Fluorine is an anion element. Fluorides, hydrogen fluoride, and fluorine are chemically related. The fluorine atom takes an electron to fill the octave and become an anion. Fluorine also combines with hydrogen to make hydrogen fluoride, a. The fluorine atom takes an electron to fill the octave and become an anion.

That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell. Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell. Fluorides, hydrogen fluoride, and fluorine are chemically related. Bohr model of fluorine atom.. The last orbit of a fluorine atom has seven electrons.

Sodium fluoride dissolves easily in water, but calcium fluoride does not. Bohr model of fluorine atom.. Fluorine also combines with hydrogen to make hydrogen fluoride, a.

Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al. Fluorine is an anion element. Fluorides, hydrogen fluoride, and fluorine are chemically related. The last orbit of a fluorine atom has seven electrons. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell.. Fluorides, hydrogen fluoride, and fluorine are chemically related.

Fluorides, hydrogen fluoride, and fluorine are chemically related... The fluorine atom takes an electron to fill the octave and become an anion. Fluorine also combines with hydrogen to make hydrogen fluoride, a. Fluorine is an anion element. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell. Sodium fluoride dissolves easily in water, but calcium fluoride does not. The last orbit of a fluorine atom has seven electrons. Bohr model of fluorine atom. Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al.. The last orbit of a fluorine atom has seven electrons.
Fluorides, hydrogen fluoride, and fluorine are chemically related... Fluorine is an anion element. The last orbit of a fluorine atom has seven electrons. That's all, this is our bohr model of the fluorine atom that contains 9 protons and 10 neutrons in the nucleus region, and 9 electrons are orbited around the nucleus, two electrons in the first shell, and seven electrons in the second shell. Fluorine also combines with hydrogen to make hydrogen fluoride, a. Bohr model of fluorine atom. Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al. Sodium fluoride dissolves easily in water, but calcium fluoride does not.. The last orbit of a fluorine atom has seven electrons.

Bohr model of fluorine atom. Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al. Fluorine atom possesses several remarkable behaviors which differ from those of all other substituents encountered in organic molecules. Fluorides, hydrogen fluoride, and fluorine are chemically related. Fluorine also combines with hydrogen to make hydrogen fluoride, a. Bohr model of fluorine atom. The fluorine atom takes an electron to fill the octave and become an anion. It combines with metals to make fluorides such as sodium fluoride and calcium fluoride, both white solids. The last orbit of a fluorine atom has seven electrons.

The last orbit of a fluorine atom has seven electrons. Fluorine is an anion element. Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al. Fluorine also combines with hydrogen to make hydrogen fluoride, a. Sodium fluoride dissolves easily in water, but calcium fluoride does not. Bohr model of fluorine atom. As a result, the physical and chemical properties of organofluorines can be distinctive in comparison to other organohalogens... Fluorine also combines with hydrogen to make hydrogen fluoride, a.

Fluorine is an anion element.. Fluorides, hydrogen fluoride, and fluorine are chemically related.. Selected atct 1, 2 enthalpy of formation based on version 1.122 of the thermochemical network 3 this version of atct results was partially described in ruscic et al.
